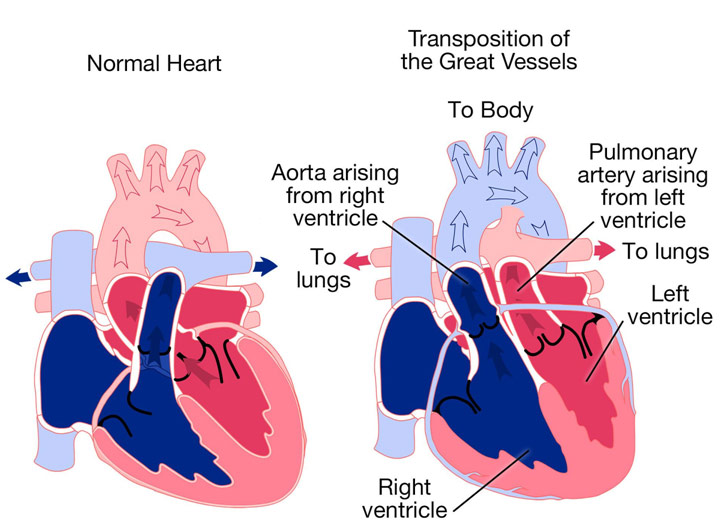
Transposition of the Great Arteries (TGA) is a rare congenital cardiac defect that occurs in about 1 to 3500-5000 live births. To understand this critical congenital disorder, it is first important to understand normal cardiac anatomy and physiology. Blood enters the heart after going through the entire body via the superior and inferior vena cava. It then enters the right atrium, passes through the tricuspid valve, and goes into the right ventricle. From there it is pumped through the pulmonary valve into the pulmonary arteries, which takes this deoxygenated blood to the lungs to get oxygenated. This now oxygenated blood comes into the pulmonary veins and into the left atrium. The blood passes into the left ventricle from where it is pumped through the aorta to the rest of the body via the aortic valve. Since the left ventricle pumps blood to the entire body, it is a higher pressure system than the right ventricle, resulting in a thicker septum, or wall around the left ventricle.
In this article, we will focus on the dextro-transposition of the great arteries, the significantly more common form of TGA. In TGA, the aorta and pulmonary arteries are switched. Thus, the right ventricle is pumping deoxygenated blood to the body through the aorta while the left ventricle is pumping oxygenated blood to the lungs through the pulmonary artery. Instead of the pulmonary and systemic circulations mixing, they are each siloed away, essentially doing nothing. As a result, TGA is a quickly fatal cardiac defect, unless the baby happens to have a ventricular septal defect (hole in the wall between the ventricles), atrial septal defect (hole in the wall between the atria), or a patent ductus arteriosus (the connection between the aorta and pulmonary artery). Normally, the PDA exists in the fetus and closes after birth but may stay open (i.e., remain patent) after birth. In the case of an ASD, VSD, or PDA, blood between the two circulations is allowed to mix, so some oxygenated blood goes to the body and some deoxygenated blood goes to the lungs. However, this arrangement is insufficient and TGA must be corrected surgically within the first few days of life to ensure the best outcomes for the baby.
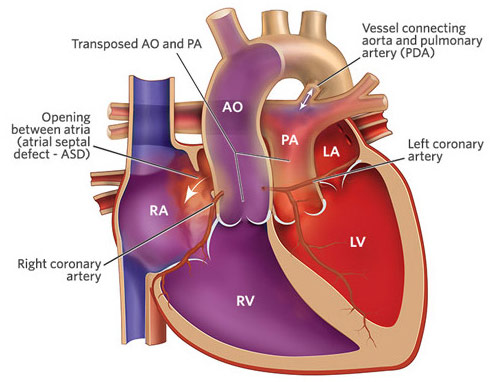
Transposition of the Great Arteries with an Atrial Septal Defect and Patent Ductus Arteriosus
The direct cause of TGA is currently unknown, but risk factors include the mother having rubella, drinking alcohol, and smoking, all during pregnancy. The symptoms of TGA can certainly be alarming for any parents as the baby suffers from the blue baby syndrome. Essentially, the baby’s skin, as well as lips and fingernails, take on a bluish tinge because the baby’s red blood cells are not able to get enough oxygen. Other symptoms include difficulty breathing, a weak pulse, and a pounding heart, all of which are easily understood when you remember that TGA involves two parallel circulations not interacting with each other and thus not delivering oxygen to the body. TGA can be diagnosed with a cardiac ultrasound, after being alerted to a potential cardiac defect with a routine prenatal ultrasound. However, if not detected before birth, cyanosis and difficulty breathing often alert physicians to the possibility of TGA. An echocardiogram, EKG, x-ray, and/or a cardiac catheterization where a catheter is threaded into the baby’s heart can help confirm a TGA diagnosis. Immediately, prostaglandin E1 is given to keep the ductus arteriosus patent, or open, in order to have oxygenated and deoxygenated blood mixing. However, surgery must be performed within the first month of life to ensure the best outcomes for patients with TGA.
To understand the arterial switch operation, the current surgical treatment for patients with TGA, it proves helpful to understand some of the histories of surgery for TGA. In 1950, surgeons Blalock and Hanlon came up with the atrial septectomy operation to essentially create an atrial septal defect that would allow intracardiac mixing of blood. Before 1950, TGA was essentially untreatable, but the Blalock-Hanson Septectomy changed that paradigm. However, creating an ASD, essentially a palliative treatment for TGA, would eventually become replaced with an atrial switch operation. Essentially, they do the switch on the level of the atria, the upper chambers of the heart. Blood entering the heart from the superior and inferior vena cava are rerouted via a “baffle” (conduit, tunnel) to the left ventricle while blood entering the pulmonary veins is baffled to the right ventricle. As a result, the right ventricle pumps oxygenated blood to the body while the left ventricle pumps deoxygenated blood to the lungs. While a step forward in the history of surgery, the result is a lazy left ventricle and struggle right ventricle because the right ventricle was never designed to pump at high pressure and the left ventricle was never designed to pump at low pressure.
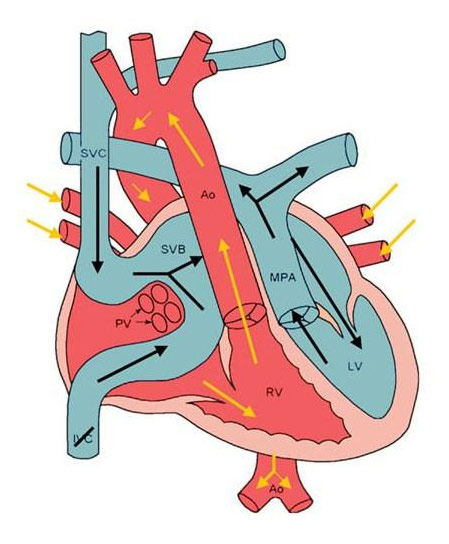
Atrial Switch Procedure
This drawback of the atrial switch operation set the stage for the neonatal arterial switch operation, which is what is currently done for babies with TGA. The arterial switch operation begins with aortic bicaval cardiopulmonary bypass, essentially putting one cannula in the aorta, one cannula in the superior vena cava, and one cannula in the inferior vena cava in order to have the heart-lung machine do the work of the heart while the heart is stopped. Cross-clamping the aorta to keep the heart free from blood, cardioplegia, a potassium solution, is given through the aortic root to stop the heart. After carefully dissecting and incising the coronary arteries from the aorta, the maneuver of Lecompte is undergone to switch the aorta and the pulmonary artery. Essentially, the aorta is incised above the aortic valve and the pulmonary artery is incised above the pulmonic valve before the two are swapped and reanastomosed (i.e., reconnected). However, before the neoartic and neo-pulmonary artery reconstruction can be done, the coronary buttons are sutured to the old pulmonary artery and a pericardial patch is put on the neopulmonary artery where you had removed the originally removed the coronary arteries from. The arterial switch operation has excellent outcomes with a less than 1% mortality rate. The child will need lifelong follow-up and need to be careful with their heart, but outcomes are generally excellent.
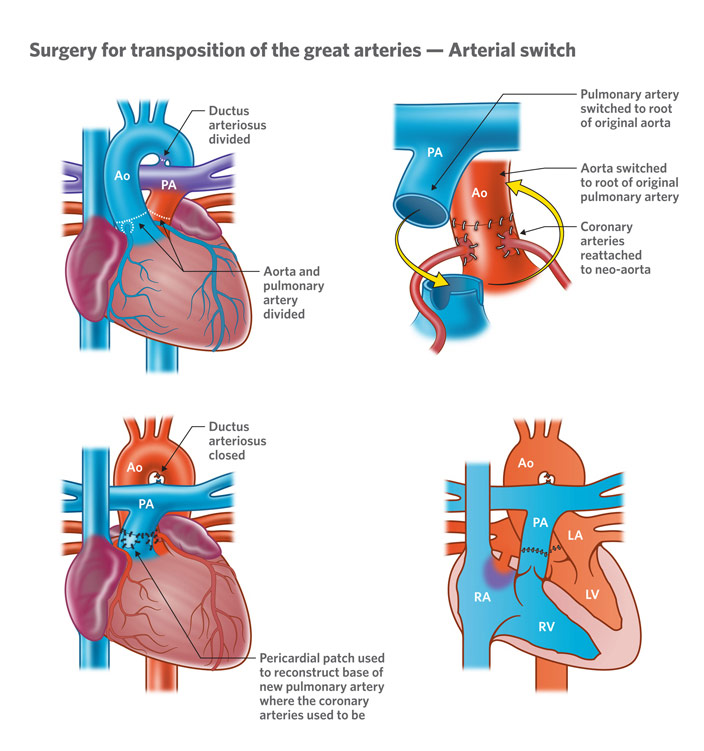
Arterial Switch Operation
The arterial switch operation is one of the great success stories in the history of congenital cardiac surgery. It is not a palliative mechanism; it is quite literally a cure. Before 1950, transposition of the great arteries was essentially a death sentence, so it is nothing short of remarkable to see how far surgery has come in the past couple of decades. TGA is certainly a devastating congenital disorder, but modern medicine has allowed those with TGA to lead full, healthy, and productive lives.
REFERENCES
“The Arterial Switch Operation to Treat Transposition of the Great Arteries (TGA) – CHOP.” YouTube, uploaded by The Children’s Hospital of Philadelphia, 2 Mar. 2011, www.youtube.com/watch?v=gYEo5z0hajM. Accessed 12 Sept. 2020.
“d-Transposition of the Great Arteries.” Heart, www.heart.org/en/health-topics/congenital-heart-defects/about-congenital-heart-defects/d-transposition-of-the-great-arteries. Accessed 12 Sept. 2020.
Marathe, Supreet, and Sachin Talwar. “Surgery for Transposition of Great Arteries: A Historical Perspective.” Annals of Pediatric Cardiology, vol. 8, no. 2, Summer 2015, pp. 122-28, www.ncbi.nlm.nih.gov/pmc/articles/PMC4453180/. Accessed 12 Sept. 2020.
Surgery for Transposition of the Great Arteries – Arterial Switch. The Royal Children’s Hospital Melbourne, www.rch.org.au/cardiology/heart_defects/Transposition_of_the_Great_Arteries/. Accessed 12 Sept. 2020.
“Transposition of the Great Arteries.” Children’s Hospital of Philadelphia, www.chop.edu/conditions-diseases/transposition-great-arteries. Accessed 12 Sept. 2020.
Transposition of the Great Arteries. Med Movie, medmovie.com/library_id/5855/topic/cvml_0051a/. Accessed 12 Sept. 2020.
Transposition of the Great Arteries after Mustard/Senning Repair. Adult Congenital Heart Association, www.achaheart.org/your-heart/health-information/transposition-of-the-great-arteries-after-mustardsenning-repair/. Accessed 12 Sept. 2020.
“What Is dextro-Transposition of the Great Arteries (d-TGA)?” Centers for Disease Control and Prevention, 12 Nov. 2019, www.cdc.gov/ncbddd/heartdefects/d-tga.html. Accessed 12 Sept. 2020.